Before we explain pTMS, let’s start with TMS. Transcranial Magnetic Stimulation (TMS) is FDA-approved, a noninvasive treatment for depression and other mental health conditions such as anxiety, PTSD, and OCD. It is also being used to explore many other brain health issues.
In many of these conditions, key brain networks responsible for mood, attention, and emotional regulation are not communicating effectively with one another. TMS uses carefully controlled magnetic pulses to safely stimulate specific brain regions and restore healthier brain network activity.
The magnetic pulses cause neurons in targeted areas of the brain to activate (usually in depression) or in other cases to slow down (usually in anxiety). Over time, repeated stimulation helps restore healthy neural connections. A helpful way to think about TMS is as physical therapy for the brain—retraining neural circuits so they can function effectively.
TMS is for clinical use since 2013 and has helped hundreds of thousands of patients worldwide.
Patients remain awake during treatment and can drive themselves to and from each session. The magnetic pulses feel like a light tapping sensation on the scalp, and the most common side effect is a mild headache that usually resolves quickly.
While traditional TMS can be effective, most clinics still locate the treatment area using a method called the “5-cm rule.” This approach estimates the stimulation site by measuring five centimeters forward from the motor cortex.
However, every brain is unique, and this estimation method can miss the most effective treatment location.
At Compassion Neurohealth, we use Precision TMS (pTMS)—an advanced and personalized approach to brain stimulation.
Precision TMS uses brain mapping, advanced analytics, and neuronavigation to identify the exact areas of the brain that need treatment for each individual patient.
Instead of estimating where to stimulate, we measure and target it.
Our approach follows five steps designed to ensure the most accurate and effective treatment possible.

We begin with advanced EEG brain mapping to measure brain activity and identify how key neural networks are functioning.
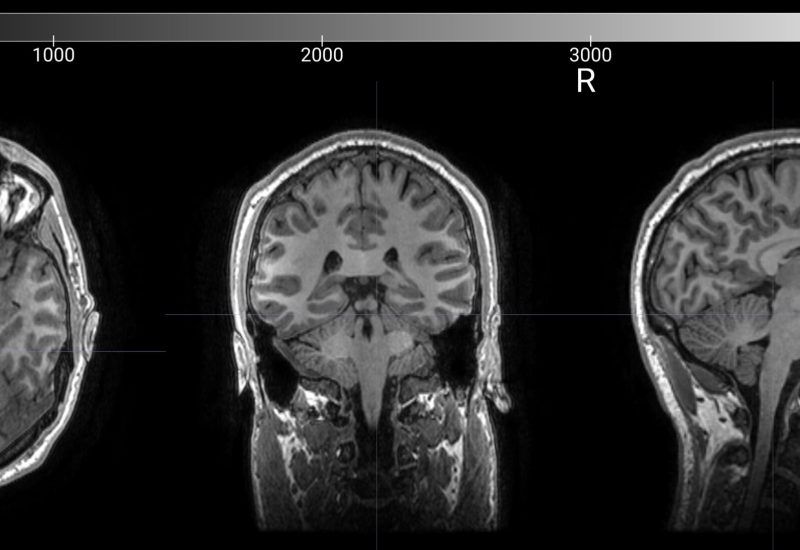
Using our proprietary HALO™ analysis platform, which evaluates brain network patterns and determines which regions are underperforming or overperforming.

We then use neuronavigation technology to precisely guide the TMS coil to the exact location identified by the HALO™. This allows us to stimulate the correct area with millimeter-level accuracy rather than relying on the traditional 5-cm rule.

Patients receive a series of TMS treatments designed to restore healthy neural connections.
Halfway through the treatment course, we re-measure brain activity to confirm the therapy is working and adjust targeting if needed.
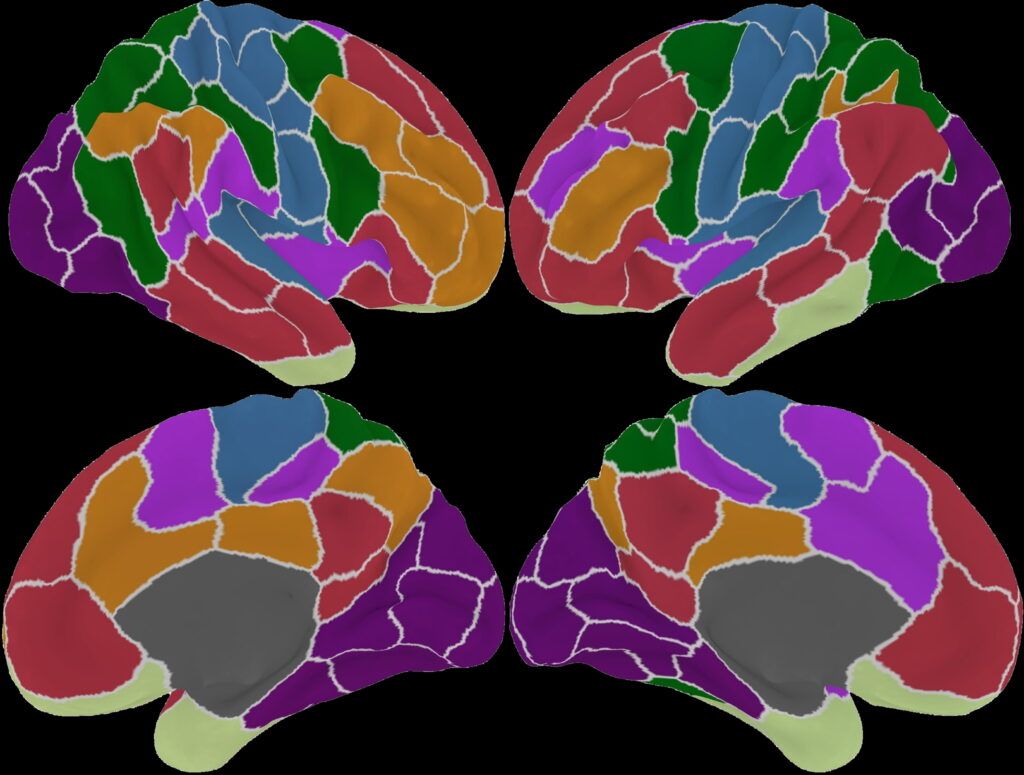
After completing treatment, we perform another brain measurement to show patients how their brain activity has changed, improved and been restored. As one patient told us “HALO validated the changes I was feeling”.
Measured. Targeted. Life-changing. Your path to hope starts here.
Precision TMS allows us to move beyond estimation and toward data-driven brain treatment.
By combining:
We are able to deliver a personalized treatment designed for each patient’s unique needs.
Our goal is simple:
Deliver the best possible outcomes in mental health care by using the most advanced brain technology available.”


TMS is safe for pregnant and breastfeeding women with no pass-through effects on your baby.
Childcare is difficult and shouldn’t be a barrier to getting TMS. You can bring your little one with you. In between TMS sessions during your visit you can rest, listen to music, nurse, play – whichever is most comfortable for you.
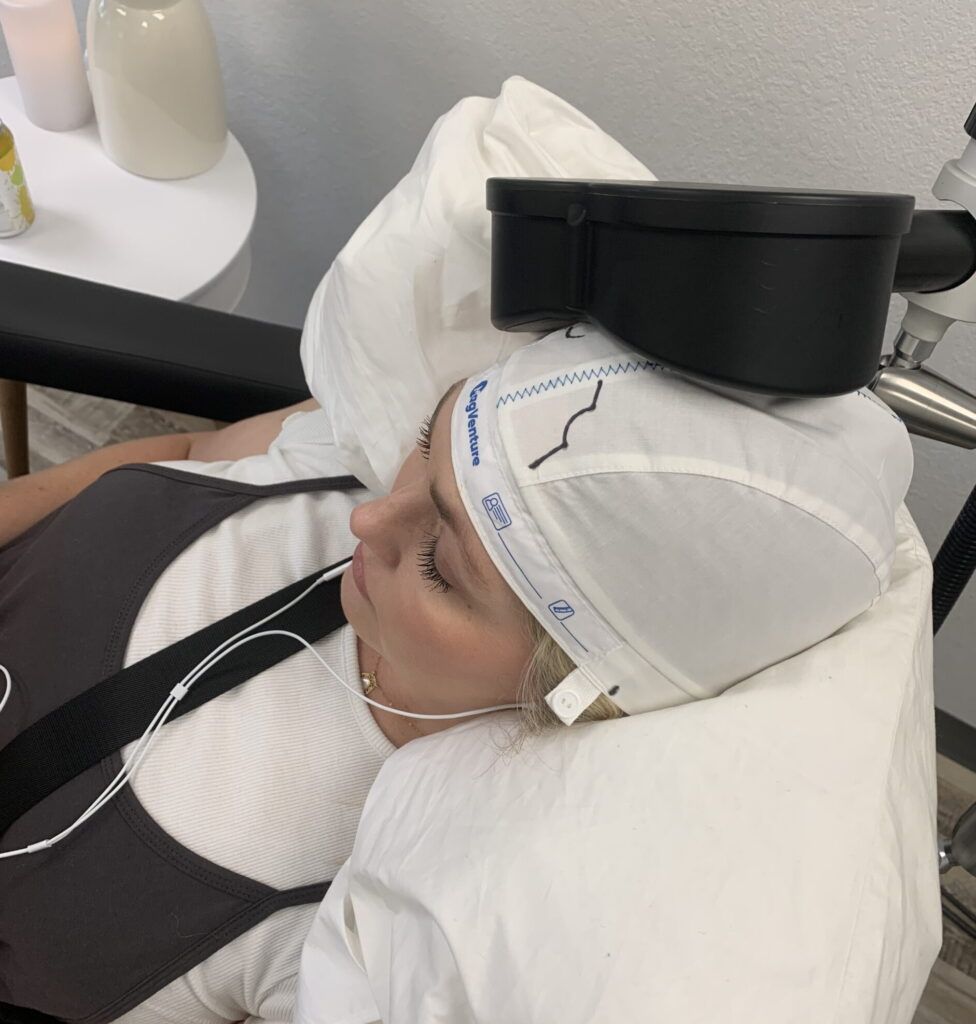
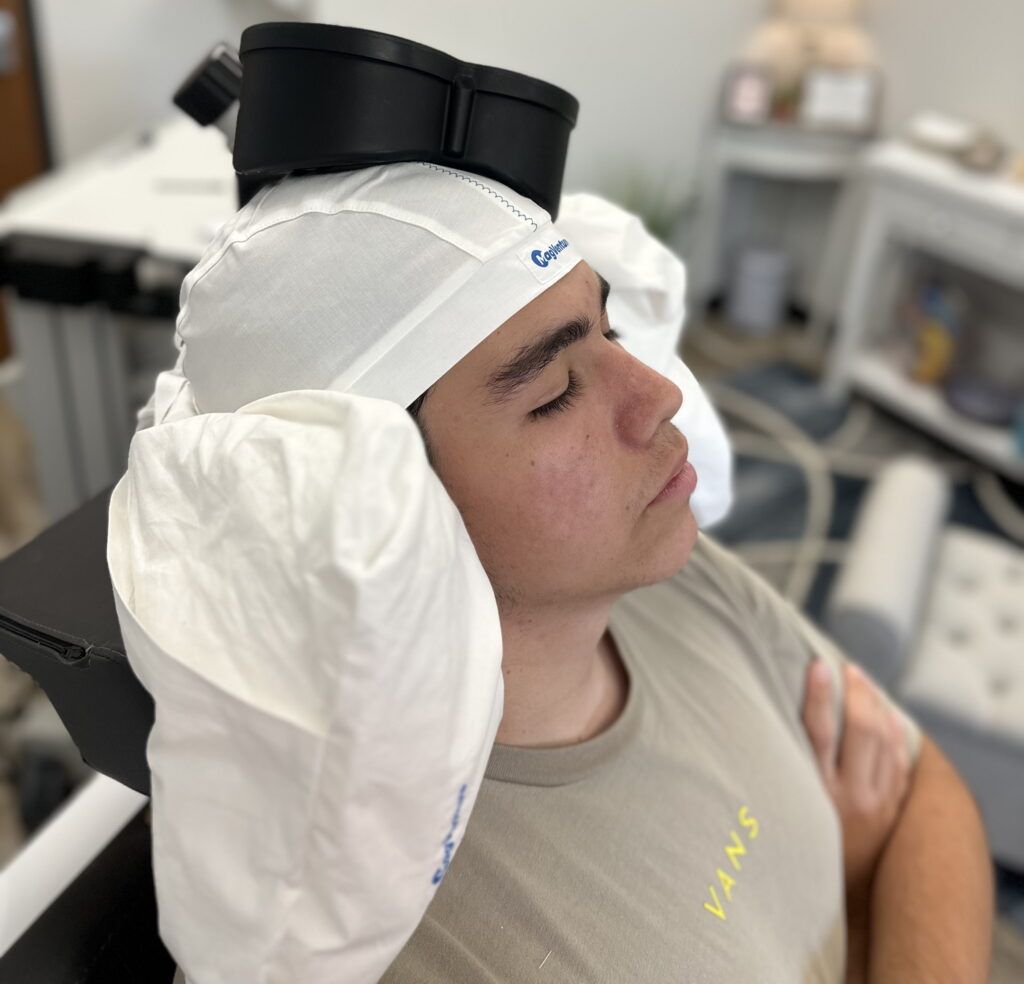
TMS is safe and effective in adolescents. The majority of patients across several clinical trials achieved clinically significant improvement in symptoms.
Now is the time to strengthen the mental health and resilience of our kids.
In a large sample of 562 adolescent patients, improvement in symptoms and remission were significantly higher in the TMS groups compared to controls, with no statistically significant difference in adverse events (side-effects). A separate review of 393 adolescent patients confirmed these findings. See FAQ below for references.
